DARKE COUNTY – The Food and Drug Administration has approved another tool in the fight against heroin and opioid addiction — a long-acting, subdermal buprenorphine implant.
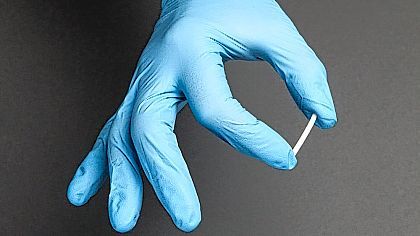
The medicated rods are implanted just beneath the surface of the skin in a single procedure and provide an ongoing release of a low dosage of buprenorphine over the span of six months. The implant is approved for patients who are already clinically stable for an least six months on other approved buprenorphine delivery systems.
“Opioid abuse and addiction have taken a devastating toll on American families. We must do everything we can to make new, innovative treatment options available that can help patients regain control over their lives,” said FDA Commissioner Robert M. Califf, M.D. “Today’s approval provides the first-ever implantable option to support patients’ efforts to maintain treatment as part of their overall recovery program.”
Buprenorphine is a medicine currently approved in the treatment of opioid addiction and is available as a tablet or film placed under the tongue or inside the cheek, both requiring that patients administer the medication to themselves every day.
The newly approved implantable form, called Probuphine, is placed under the skin in the upper arm in an outpatient setting and is removed the same way at the end of the treatment period. Where tablets and films can be forgotten, lost or stolen, the implant eliminates these risks to ensure steady, reliable administration of the needed drug.
Other medications used in treatment of opioid addiction include methadone and naltrexone.
Medication-assisted treatment (MAT) has been proven to help patients recover from opioid addiction, reduce fatal overdoses, improve social functioning, reduce criminal activity, and lessen the risk of transmitting infectious diseases, such as HIV and hepatitis C.
Jerri Lynn Stanley, Clinical Director for Recovery and Wellness Center in Greenville, said the organization has been using Vivitrol (naltrexone) for about a year now, and “It is going well.” Patients typically begin on the monthly shot, along with counseling and other support, and after a period of time – usually about six months – they can move on to taking the daily pills themselves.
The new implants could become the next step in the process, as a maintenance tool, until medication can be eliminated from the recovery process.
Expanding the use and availability of medication-assisted treatment (MAT) options like buprenorphine is an important component of the FDA’s opioid action plan, and one of three top priorities for the U.S. Department of Health and Human Services’ Opioid Initiative.
MAT is a comprehensive approach that combines approved medications with counseling and other behavioral therapies to treat patients with opioid use disorder. Regular adherence to MAT with buprenorphine reduces opioid withdrawal symptoms and the desire to use, without causing the cycle of highs and lows associated with opioid misuse or abuse. At sufficient doses, it also decreases the pleasurable effects of other opioids, making continued opioid abuse less attractive. According to the Substance Abuse and Mental Health Services Administration, patients receiving MAT for their opioid use disorder cut their risk of death from all causes in half.
“Scientific evidence suggests that maintenance treatment with these medications in the context of behavioral treatment and recovery support are more effective in the treatment of opioid use disorder than short-term detoxification programs aimed at abstinence,” said Nora Volkow, M.D., director of the National Institute on Drug Abuse at the National Institutes of Health. “This product will expand the treatment alternatives available to people suffering from an opioid use disorder.”
Probuphine consists of four, one-inch-long rods that are implanted under the skin on the inside of the upper arm and provide treatment for six months. If further treatment is needed, new implants may be inserted in the opposite arm for one additional course of treatment. The FDA is requiring postmarketing studies to establish the safety and feasibility of placing the Probuphine implants for additional courses of treatment.